 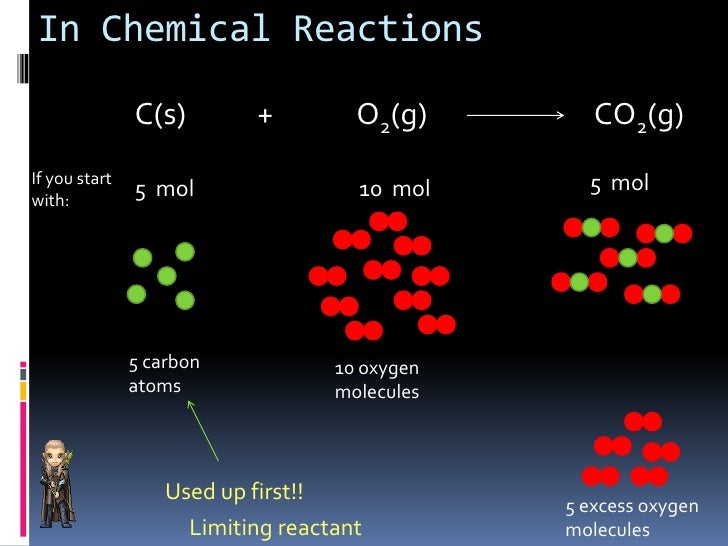
The short answer is because H 2 and O 2 react with different mole ratio: However, when it comes to determining the limiting reactant (LR) in chemical reactions, it is slightly different as we cannot determine the limiting reactant just by looking at the initial amounts of the reactants.įor example, if we mix together 6 moles of hydrogen gas and 4 moles of oxygen gas, we cannot say that the oxygen is the limiting reactant even though yes, it is present in less quantity. The same situation occurs in chemical reactions, where one of the reactants is less than the other one and there is not enough of it to consume/react with all the second reactant. We would all agree that the number of chairs or simply the chair is the limiting factor in this situation since there is simply not enough of them. What is the limiting reactant? Let’s consider a situation where five people need to take a seat but there are only four chairs available. Transcript of the video and more practice problems below 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
